Chapter 35 Steroid Ineffective for Prevention
Transient Corticosteroid Treatment Permanently Amplifies the Th2 Response in a Murine Model of Asthma
By RE. Wiley et al. published in the J Immunol. 2004 Apr 15;172(8):4995-5005
This is the animal experimental study for asthma. Though this is not a model for atopic dermatitis (AD), it shows very interesting data concerning the relationship between local (inhaled) administration of corticosteroid and Th2 activation.
To sum up the conclusion, this study warns that routinely suppressing a mild asthma with steroid inhalant will not prevent the future aggravation of symptoms, or rather might induce it.
In the experiment, mice were sensitized to OVA (ovalbumin) through inhalation.
Transient Corticosteroid Treatment Permanently Amplifies the Th2 Response in a Murine Model of Asthma
By RE. Wiley et al. published in the J Immunol. 2004 Apr 15;172(8):4995-5005
This is the animal experimental study for asthma. Though this is not a model for atopic dermatitis (AD), it shows very interesting data concerning the relationship between local (inhaled) administration of corticosteroid and Th2 activation.
To sum up the conclusion, this study warns that routinely suppressing a mild asthma with steroid inhalant will not prevent the future aggravation of symptoms, or rather might induce it.
In the experiment, mice were sensitized to OVA (ovalbumin) through inhalation.
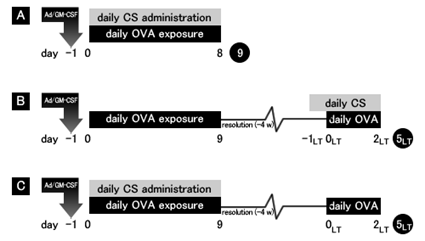
The above A, B and C experimental systems were used.
A: Two groups of mice were sensitized to OVA through inhalation for 8 days, during which time one group was made to inhale CS (corticosteroid) at the same time and the other was not. The two groups were compared on Day 9 to examine the difference in the outcome between the group treated with CS during sensitization and the group untreated.
B: The two groups of mice were sensitized to OVA through inhalation for 8 days. Then, after a 4- week interval, when inflammation subsided enough, mice were exposed to OVA again, which caused stronger inflammation than before as sensitization had been established. Then, comparison was made between the group treated with CS during the second exposure and the group untreated.
C: Two groups of mice were sensitized to OVA through inhalation for 8 days, during which time one group was made to inhale CS (corticosteroid) at the same time and the other was not. Then, after a 4- week interval, when inflammation subsided enough, mice were exposed to OVA again with both groups not being treated with steroid to see the outcome.
Let’s look at the outcome of Experimental System A.
A: Two groups of mice were sensitized to OVA through inhalation for 8 days, during which time one group was made to inhale CS (corticosteroid) at the same time and the other was not. The two groups were compared on Day 9 to examine the difference in the outcome between the group treated with CS during sensitization and the group untreated.
B: The two groups of mice were sensitized to OVA through inhalation for 8 days. Then, after a 4- week interval, when inflammation subsided enough, mice were exposed to OVA again, which caused stronger inflammation than before as sensitization had been established. Then, comparison was made between the group treated with CS during the second exposure and the group untreated.
C: Two groups of mice were sensitized to OVA through inhalation for 8 days, during which time one group was made to inhale CS (corticosteroid) at the same time and the other was not. Then, after a 4- week interval, when inflammation subsided enough, mice were exposed to OVA again with both groups not being treated with steroid to see the outcome.
Let’s look at the outcome of Experimental System A.
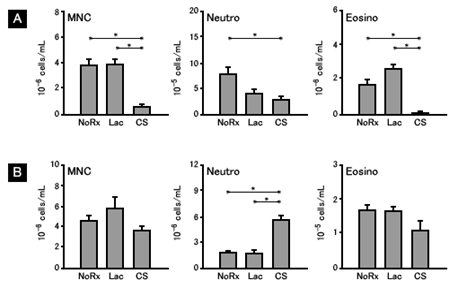
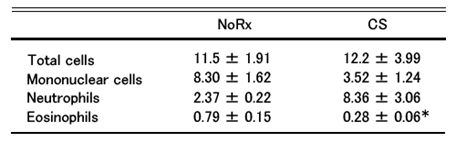
Mononuclear cell (NMC), neutrophil (Neutro) and eosinophil (Eosino) in alveolar lavage fluid (A) and in peripheral blood (B) were compared between the steroid administered (treated) group (CS) and untreated control group (NoRx and Lac). The treated group had less inflammatory cells in the alveolar lavage fluid and increased number of neutrophils in the peripheral blood. It is well known that steroid administration increases the number of neutrophils in peripheral blood and this is the result consistent with clinical experience.(These are just the confirmation of what is already known.)
Next, cytokines in alveolar lavage fluid (A) and OVA specific IgE in serum were measured. In the treated group, Th2 cytokine and OVA specific IgE levels increased. (Interesting results that attract attention as a prologue.)
Let’s look at Experimental System B now.
Let’s look at Experimental System B now.
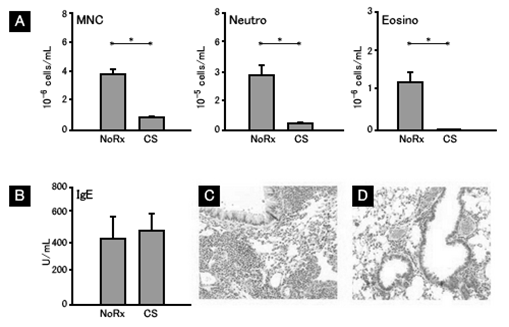
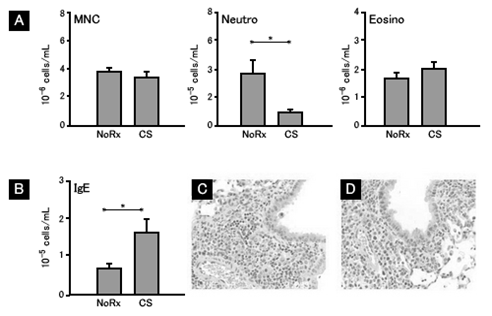
Mononuclear cells (NMC), neutrophil (Neutro) and eosinophil (Eosino) in peripheral blood (B) of the treated group (CS) are apparently less in number than those of the control group (NoRx). As IgE, there is little difference between the two groups. Looking at the pathological tissue of the lung, inflammation is strong for the control group (C) and mild for the treated group (D). (Interesting results that raise expectations for what will come next.)
Eosinophils in peripheral blood dropped in the treated group. (It’s natural.)
Let’s look at Experimental System C now.
Let’s look at Experimental System C now.
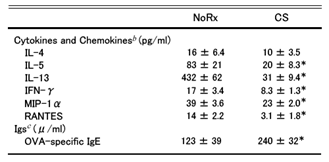
After sensitization and re-exposer to OVA, cells of intrathoracic lymph nodes taken from the control group (NoRx) and the treated group (CS) were cultured and its supernatant was measured for cytokines. As to the medium, nothing was added to the medium. As to OVA, the cultured cells were stimulated with OVA. Th2 cytokines such as IL-4, 5, 13 significantly increased in the treated group.
In the treated group for which steroid was administered simultaneously during allergen sensitization, IgE in the peripheral blood significantly increased. As to the pathological tissue of the lung, it looks almost the same in the control group (C) and the treated group (D). (Inflammation is equally strong in both groups. This is the key point, which indicates that steroid administration, as a treatment, during allergen sensitization will enhance Th2 response, which lead to excessive production of IgE when re-exposed to allergens later.
In contrast to Experimental System B, Eosinophils also increased in the CS administered group.
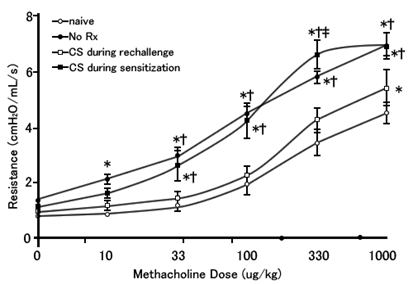
The above graph shows the pulmonary resistance in each group of naïve (not sensitized), NoRx (sensitized with no CS administration), CS during rechallenge (CS administered during re-exposure to allergen), and CS during sensitization (CS administered during sensitization). The lines for the CS during sensitization and the NoRx groups almost overlap. That is, treatment with steroid during sensitization has little effect of alleviating symptoms that develop when re-exposed to allergens later, or rather may enhance the Th2 response to increase the production of IgE or eosinophils and make the allergy intractable.
This is the study on treatment with steroid when the lung is exposed to allergens through inhalation and cannot be directly applied to the cases of atopic dermatitis, which is the skin disorder. But as an immunologic response review, it can be highly applicable to the skin. If applied to the skin, for example, it can be said that IgE antibody or eosinophil level goes up when steroid is topically applied to a baby who is being sensitized to environmental allergens such as house dust.
Some doctors say, “A rash needs to be suppressed with steroid while it is mild in symptoms, or it will get worse and chronic.” But this might be opposite in a real sense.
This is a purely immunological episode, differing from Dr. Cork’s barrier disruption theory, and cannot explain the mechanism of steroid addiction that leads to rebound. I have not heard of asthma patients who got addicted to steroids and had a hard time to withdraw from steroids. Steroid addiction or rebound is a story peculiar to the skin exposed to topical steroids for a long time. Therefore, Dr. Cork’s theory, that implicated impaired epidermal barrier as a causative reason, is persuasive.
This study does not explain the mechanism of steroid addiction but warns that steroid application may make an allergy intractable.
This is the study on treatment with steroid when the lung is exposed to allergens through inhalation and cannot be directly applied to the cases of atopic dermatitis, which is the skin disorder. But as an immunologic response review, it can be highly applicable to the skin. If applied to the skin, for example, it can be said that IgE antibody or eosinophil level goes up when steroid is topically applied to a baby who is being sensitized to environmental allergens such as house dust.
Some doctors say, “A rash needs to be suppressed with steroid while it is mild in symptoms, or it will get worse and chronic.” But this might be opposite in a real sense.
This is a purely immunological episode, differing from Dr. Cork’s barrier disruption theory, and cannot explain the mechanism of steroid addiction that leads to rebound. I have not heard of asthma patients who got addicted to steroids and had a hard time to withdraw from steroids. Steroid addiction or rebound is a story peculiar to the skin exposed to topical steroids for a long time. Therefore, Dr. Cork’s theory, that implicated impaired epidermal barrier as a causative reason, is persuasive.
This study does not explain the mechanism of steroid addiction but warns that steroid application may make an allergy intractable.